Capsule Sponge Test
Simple Clinic Test Offers Alternative to Endoscopy when Screening for Barrett’s Oesophagus.
Enquire about the Capsule Sponge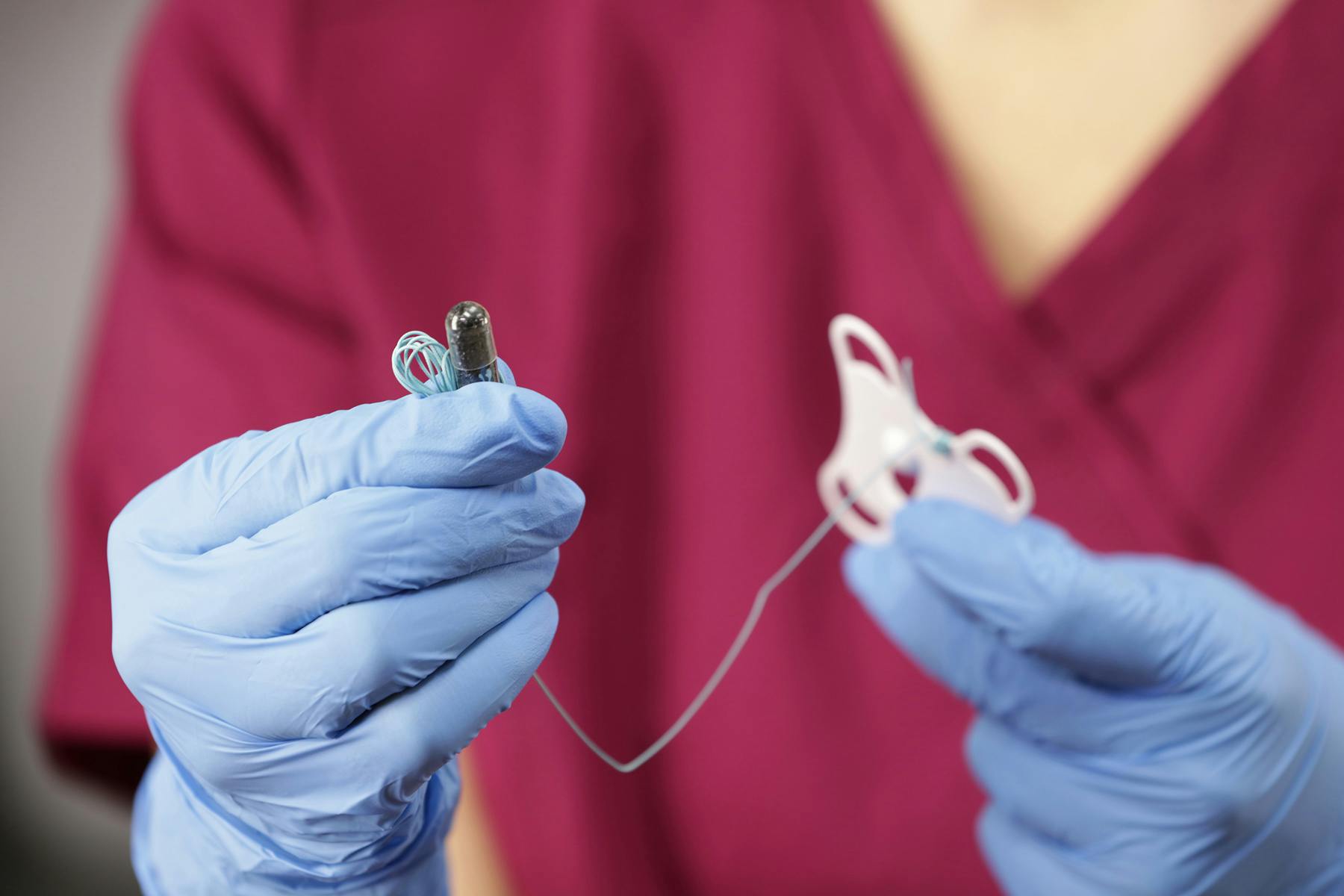
What is a Capsule Sponge Test (or 'Sponge On A String' test) for?
The Capsule Sponge Test is a cutting-edge screening tool designed to detect Barrett's Oesophagus and oesophageal cancer early, and reduce the likelihood of either developing. Unlike traditional endoscopy, which is invasive, uncomfortable and often requires sedation, the Capsule Sponge Test (sometimes known by brand names such as EndoSign or Cytosponge) is simple, quick, and minimally invasive.
How does it work?
- Swallowing the Capsule: The test begins with the patient swallowing a small capsule attached to a thin string. The capsule contains a compressed sponge.
- Sponge Expansion: After around 5 minutes, the capsule dissolves in the stomach, and the sponge expands.
- Retrieving the Sponge: The sponge is then gently pulled back up through the oesophagus by the string, collecting cells from the oesophageal lining as it moves.
- Analysis: The collected cells are sent to a laboratory where they are analysed for signs of Barrett's Oesophagus
Benefits of the Capsule Sponge Test?
Non-Invasive: No need for sedation or the discomfort associated with endoscopy.
Quick and Convenient: The entire process takes less than 20 minutes.
Accurate and Reliable: Proven to be highly effective in detecting Barrett's Oesophagus, as well as oesophageal cancer and eosinophilic oesophagitis.
Cost-Effective: Lower cost compared to the traditional method of gastroscopy, making it accessible to more patients.
Who should consider the Test?
Individuals with a diagnosis of GERD.
Those with a family history of Barrett's Oesophagus or oesophageal cancer.
Patients experiencing persistent heartburn or reflux symptoms.
People at higher risk of developing Barrett’s Oesophagus. This includes the following risk factors:
- Males
- Caucasians
- Obesity
- Age over 40
Early detection can make a significant difference in outcomes, and this test offers a practical solution for proactive health management.
Take charge of your health and get in touch
RefluxUK are the largest and most specialised centre for the diagnosis and treatment of reflux and other benign upper GI disorders in the UK.
Don't wait for symptoms to worsen. The Capsule Sponge Test provides a proactive approach to managing your oesophageal health.
For more information about the Capsule Sponge Test for Barrett's Oesophagus, or to schedule a consultation, please contact us by submitting your enquiry on the link below.
Our Capsule Sponge Kits are provided by EndoSign®, who are supported by the University of Cambridge, Cancer Research UK and Heartburn Cancer UK.
